

| |

History
of Malaria in Europe during the Middle Ages:
From Shakespeare to Defoe: Malaria in England in
the Little Ice Age
Paul Reiter
Centers for Disease Control and Prevention, San Juan, Puerto Rico

| Present global
temperatures are in a warming phase that began 200 to 300
years ago. Some climate models suggest that human activities
may have exacerbated this phase by raising the atmospheric
concentration of carbon dioxide and other greenhouse gases.
Discussions of the potential effects of the weather include
predictions that malaria will emerge from the tropics and
become established in Europe and North America. The complex
ecology and transmission dynamics of the disease, as well as
accounts of its early history, refute such predictions. Until
the second half of the 20th century, malaria was endemic and
widespread in many temperate regions, with major epidemics as
far north as the Arctic Circle. From 1564 to the 1730s—the
coldest period of the Little Ice Age—malaria was an
important cause of illness and death in several parts of
England. Transmission began to decline only in the 19th
century, when the present warming trend was well under way.
The history of the disease in England underscores the role of
factors other than temperature in malaria transmission. |
The earth's climate has always been in a state of change. The past
250 to 300 years have seen a fairly steady warming trend. Average
temperatures are now approaching those at the height of the Medieval
Warm Period, near the end of the 12th century. The intervening centuries
included a much colder period, the Little Ice Age, by far the most
important climatic fluctuation in recent history (1).
Such fluctuations, spanning several generations, are natural phenomena
that have recurred several times in the past 10,000 years. They take
place against a backdrop of episodes of longer duration and greater
impact, such as the last Ice Age (1,600,000 to 10,000 years ago). In
recent years, there has been growing concern that human activities may
be modifying the natural climate. A decline in temperatures from the
1940s to the late 1970s gave rise to warnings that industrial pollutants
were causing global cooling (2,3).
Subsequent warming has been attributed to increased concentrations of
atmospheric carbon dioxide produced by burning fossil fuels, and other
greenhouse gases (4). Climate models suggest that this
trend could accelerate in the coming century, although the contribution
of human-induced greenhouse gases to global temperatures is far from
clear (4-6).
Discussions of the potential impact of human-induced global warming
frequently include malaria, a disease widely perceived as tropical.
Articles in the popular and scientific press have predicted that warmer
temperatures will result in malaria transmission in Europe and North
America (7-12). Such predictions,
often based on simple computer models, overlook malaria's history; until
recently, malaria was endemic and common in many temperate regions, and
major epidemics extended as far north as the Arctic Circle (13).
Despite the disappearance of the disease from most of these regions, the
indigenous mosquitoes that transmitted it were never eliminated and
remain common in some areas. Thus, although temperature is important in
the transmission dynamics of malaria, many other variables are of equal
or greater importance. This article reviews the history of the disease
in a nontropical country—England—during the coldest years of the
Little Ice Age.
The Little Ice Age
The 16th century is the first period for which we have a reliable
history of climate and weather. Private diaries, ships' logs, accounts
of military campaigns, and similar sources give descriptions of wind
direction, wind speed, cloud formations, and other weather indicators.
Precisely dated annals, chronicles, audited accounts, agricultural
records, tax ledgers, and other archival material provide indirect
information, particularly on extreme weather events, such as droughts,
floods, or unusual cold. Additional evidence is available from glacial
moraines, lake and ocean sediments, pollen strata, deposits of insects,
tree rings, coral structure, radiometric analysis of ice cores,
archaeologic sites, and many other sources. All this information can be
combined to reconstruct past climates (Figure 1).
|
Figure
1 |
|
|
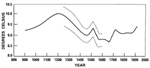 |
|
|
Click
to view enlarged
image
Figure 1. Estimated mean annual temperatures (°C) prevailing in
central England since A.D. 800. These are 50-year averages,
based on a variety of proxy data (see text). Dotted lines
indicate the range of apparent uncertainty of the derived
values.
|
|
The first half of the 16th century appears to have been much warmer
than the previous 150 years, which had seen a fairly steady decline in
temperatures after the Medieval Warm Period. However, the new genial
climate was not to last. In the middle of the century, after a decade or
so of exceptionally warm years—warm enough for people to bathe in the
Rhine in January—a drastically sharp change occurred. The winter
1564-65 was long and bitterly cold and was the first of many that
brought great hardship and strife throughout Europe. Climatologists
regard the next 150 to 200 years the climax of the Little Ice Age. On
average, temperatures were probably lower than in any similar episode
since the end of the last major Ice Age (14).
The impact of this sudden change of climate was dramatic and global (1).
Glaciers advanced rapidly in Greenland, Iceland, Scandinavia, and the
Alps. The Arctic pack ice extended so far south that there are six
records of Eskimos landing their kayaks in Scotland. Large tracts of
land, particularly at higher latitudes and altitudes, had to be
abandoned. In many years, snowfall was much heavier than recorded before
or since, and the snow lay on the ground for many months longer than it
does today. Many springs and summers were outstandingly cold and wet,
although there was great variability between years and groups of years.
Crop practices throughout Europe had to be altered to adapt to the
shortened, less reliable growing season, and there were many years of
dearth and famine. Violent storms caused massive flooding and loss of
life. Some of these resulted in permanent losses of large tracts of land
from the Danish, German, and Dutch coasts. The dramatic cooling was
captured in the paintings of the Flemish artist Pieter Bruegel
(1525–1569), who initiated a new genre by completing at least seven
winter landscapes in 2 years (Figure 2).
|
|
Climate changes were equally striking in other parts of the world. In
Ethiopia and Mauritania, permanent snow was reported on mountain peaks
at levels where it does not occur today. Timbuktu, an important city on
the trans-Saharan caravan route, was flooded at least 13 times by the
Niger River; there are no records of similar flooding before or since.
In China, warm weather crops, such as oranges, were abandoned in Kiangsi
Province, where they had been grown for centuries. In North America, the
early European settlers also reported exceptionally severe winters. For
example, in 1607-8 ice persisted on Lake Superior until June (1).
Ague
The identity of diseases described in early literature is often
uncertain, but fevers with a periodicity described as tertian or quartan
are highly suggestive of malaria. Descriptors in the context of such
accounts are often supportive. For example, in temperate regions,
epidemic transmission of malaria tended to occur in late summer and
autumn, giving rise to the common terms aestivo-autumnal or harvest
fever for tertians and quartans.
During the Medieval Warm Period, mention of malarialike illness was
common in the European literature from Christian Russia to caliphate
Spain: "As one who has the shivering of the quartan so near,/ that
he has his nails already pale/ and trembles all, still keeping the
shade,/ such I became when those words were uttered." (The Inferno,
Dante [1265-1321]).
The English word for malaria was ague, a term that remained in common
usage until the 19th century. The Medieval Warm Period was already on
the wane when Geoffrey Chaucer (1342-1400) wrote, in the Nun's Priest's
Tale, "You are so very choleric of complexion./ Beware the mounting
sun and all dejection,/ Nor get yourself with sudden humours hot;/ For
if you do, I dare well lay a groat/ That you shall have the tertian
fever's pain,/ Or some ague that may well be your bane."
Such mention of agues did not disappear when the coldest years of the
Little Ice Age began. In 16th century England, many marshlands were
notorious for their ague-stricken populations and remained so well into
the 19th century. William Shakespeare (1564–1616), who was born in the
autumn of Bruegel's first fierce winter, mentioned ague in eight of his
plays. For example, in The Tempest (Act II, Scene II), the slave Caliban
curses Prosper, his master: "All the infections that the sun sucks
up/ From bogs, fens, flats, on Prosper fall and make him / By inch-meal
a disease!" Later, Caliban is terrified by the appearance of
Stephano, who, mistaking his trembling and apparent delirium for an
attack of malaria, tries to cure the symptoms with alcohol: ". . .
(he) hath got, as I take it, an ague . . . he's in his fit now and does
not talk after the wisest. He shall taste of my bottle: if he have never
drunk wine afore it will go near to remove his fit . . . Open your
mouth: this will shake your shaking . . . if all the wine in my bottle
will recover him, I will help his ague."
Alcohol and opium were commonly used to suppress the rigors of the
first stage of the malarial paroxysm (15). We can
assume that Shakespeare's audience was familiar with such details,
although the play, one of his last, was probably not presented until
1611, long after the change to a colder climate.
Geographic Distribution: the Marsh Parishes
Five indigenous species of Anopheles mosquito are capable of
transmitting malaria in England. The most competent, An. atroparvus,
prefers to breed in brackish water along river estuaries. Contemporary
accounts of the distribution of ague in 16th and 17th century England
reflect the ecology and distribution of this species. For example, the
anaerobic bacterial flora of saline mud produces a strong and
distinctive sulfurous odor. This was widely perceived to be the actual
cause of agues in salt marsh areas—Shakespeare's "unwholesome
fens"—hence the Italian term mala aria (bad air).
The history of malaria in England and its demographic, epidemiologic,
and social impact have been described in detail (16).
Descriptions by 17th century topographers of the airs and waters
surrounding settlements along the River Medway (a tributary of the
Thames estuary) alluded to this association (17).
Thus, Upchurch, a town on the open estuary: "lies in a most
unhealthy situation, close to the marshes . . . the noxious vapours
arising from which subject the inhabitants to continued intermittents."
In nearby Iwade: " . . . in summer dry weather, the stench of the
mud in the ponds and ditches. . . contribute so much to its
unwholesomeness, that almost everyone is terrified to live in it . .
." In Burnham, further upstream, the air was: "rather more
healthy . . . owing to the marshes being fewer and less offensive than
those lower down the river. . ." and finally, the freshwater
reaches were "far more healthy . . . instead of the noisesome
smells, arising from the salt marshes . . . the river here is
encompassed with a range of pleasant fertile meadows, greatly conducive
both to health and pride."
Even vicars of the Church were afraid of living in the estuarine
areas. Dobson (15) reviewed several questionnaire
surveys conducted by their bishops that asked "Do you reside
personally upon your Cure, and in your Parsonage House? If not, where do
you reside? What is the reason for your nonresidence?"
Nearly all the vicars of coastal "marsh parishes" answered
no to the first question (Dobson lists 28 such parishes from the
counties of Essex and Kent), and gave reasons such as "the place is
so very Agueish," "frequently taken with agues and
fevers," "the Thames having a very foul shore in this parish .
. . attacked by so many repeated agues," "so violently
afflicted with the worst of agues and languishing so long under it till
our constitutions were almost broke," and "so unhealthy a
situation as to be absolutely unfit for any curate."
Demographic data give striking justification for the vicars' fears (16)
(Figures 3 and 4, Table).
Descriptions of the marsh inhabitants resemble those of malaria-endemic
area populations in the tropics today. Visitors commented on the swollen
bellies of the children and their sallow, sickly faces, suggestive of
anemia. An enlarged spleen was called ague cake. Nevertheless, despite
the coldness of the climate, there is no indication that there was any
major change in these death rates during the 17th century.
Not all the summers of the 17th century were equally cool. The
diarist Samuel Pepys (1633-1703), who himself had ague, wrote that the
summers of 1661, 1665, and 1666 were dry and remarkably hot. This may
well have enhanced transmission rates, for Thomas Sydenham described an
epidemic of tertian and some quartan fevers in 1661, which "was
doing frightful mischief" by August. Drought malaria, common in
many parts of the world, arises when rivers and ponds are reduced to the
smaller pools and puddles that anopheline mosquitoes prefer for
breeding. Dobson (15) found a positive correlation
between warm, dry summers in 1660 to 1810 and seasonal burial rates in
Bradwell-juxta-Mare, a marsh parish in Essex. Macdonald (18)
showed a strong relationship between high summer temperatures and cases
of ague in Kent in the mid-19th century. High was defined as days when
the temperature rose above 16°C, as measured at the Royal Greenwich
Observatory. Such hot weather, though clearly not tropical, could
certainly have increased the probability of transmission by shortening
the extrinsic incubation period (the time required for the mosquito to
become infective after feeding on an infected person). Nevertheless,
agues were not restricted to the warmer years.
Clinical Descriptions
Some accounts of intermittent fevers during the Little Ice Age were
by physicians whose emphasis on precise methods, detailed observations,
and accurate records leave us in no doubt that they were referring to
malaria. William Harvey (1578-1657) made careful pre- and postmortem
observations of cases of ague in London hospitals (St. Bartholomew's and
St. Thomas').1 His interest was
probably sharpened because a persistent ague had caused him to miss much
of his final year (1598-99) at the University of Cambridge. In his
treatise on the circulation of the blood Exercitatio Anatomica de Motu
Cordis et Sanguinis in Animalibus (1628), he gave a detailed description
of the clinical pathologic features of the febrile episodes: "In
tertian fever . . . in the first instance . . . the patient (is)
short-winded, disposed to sighing, and indisposed to exertion . . . the
blood (is) forced into the lungs and rendered thick. It does not pass
through them (as I have myself seen in opening the bodies of those who
had died in the beginning of the attack), when the pulse is always
frequent, small, and occasionally irregular; but the heat increasing . .
. and the transit made, the whole body begins to rise in temperature,
and the pulse becomes fuller and stronger. The febrile paroxysm is fully
formed . . ."
Obstruction of circulation—commonly termed sludging—is mainly
seen in Plasmodium falciparum infections. It results from complex
changes in the consistency of the blood (19) and is
manifest in many organs, although most often cited in the context of
cerebral malaria. As Harvey noted, this dangerous condition often occurs
suddenly, soon after the initial symptoms of a paroxysm.
|
| |
|
|
Table. Deaths per
1,000 per year in late 18th century England
(17) and 20th century Nigeria (39)
|
|

|
| |
Essex, England
|
Garki, Nigeria
|
|
Age
|
Nonmarsh
|
Marsha |
Age |
Savanna |
|

|
|
5
|
44
|
95.3 |
1-4 |
154 |
|
6-10
|
6.3
|
9.4 |
5-8 |
15 |
|
11-15
|
6.8
|
10.8 |
9-18 |
10 |
|
16-20
|
8.4
|
12.7 |
19-28 |
6 |
|

|
|
a The high death
rates in the marsh parishes are attributable to
malaria and were actually lower than in the previous century
during the peak of the Little Ice Age. Moreover, the Essex
data are skewed because many of the marsh
communities included predominantly men. The
rate was similar to that measured in the
1970s by the World Health Organization
in a malaria-endemic area of sub-Saharan Africa.
|
|
| |
Thomas Sydenham (1624-1689) is widely regarded as a founder of
clinical medicine and epidemiology. His book on fevers (1666), which was
later expanded into Observationes Medicae (1676), a standard text for 2
centuries, includes extensive details of agues, with descriptions of the
course of paroxysms, the periodicity of tertians versus quartans, and
the seasonality of the disease. As with Harvey, there can be no doubt
that his descriptions refer to malaria; moreover, he states, "When
insects do swarm extraordinarily and when . . . agues (especially
quartans) appear early as about midsummer, then autumn proves very
sickly." We cannot assume that insects referred to mosquitoes, but
the statement that an early appearance of quartans was a premonition of
a high incidence of illness (presumably agues?) has the distinct ring of
modern epidemiology. Nevertheless, it is clear from these and other
sources that the disease remained prevalent, even in the coldest years.
For example, Harvey's ague began in the fall of 1597, yet the years 1594
to 1597 had been so cold and wet that wheat harvests were a disaster (1).
Similarly, in 1657 to 1658, snow lay on the ground for 102
days—indicating exceptionally cold weather even for the times. The
summer crops were a disaster, yet Oliver Cromwell (1599-1658) died of a
tertian ague in September 1658, just as another severe winter was
setting in.
Malaria Species
P. vivax and P. falciparum both have a tertian
periodicity, so it is unclear which species was responsible for tertiary
ague in England. P. vivax can persist for many years as a dormant
hypnozoite in the liver, giving rise to occasional clinical relapses
(caused by production of the blood stages of the parasite), whereas
there is no evidence of hypnozoites in P. falciparum.
Twentieth-century studies in Russia and Holland showed that some strains
of P. vivax from the northern hemisphere—given the subspecific
name P. vivax hibernans—did not produce clinical symptoms until
8-9 months after the infective bite. Both features would clearly have
survival value for P. vivax in a temperate climate, enabling it
to cope with long winters and episodes of successive cold summers. On
the other hand, P. vivax is generally a more benign pathogen than
P. falciparum, but the death rate in the English marsh parishes
suggests that infections were far from benign. This may indicate that P.
falciparum was also present, and Harvey's observations on the
consistency of blood in fatal cases support this possibility.
Laboratory studies have shown that tropical strains of P.
falciparum do not multiply in European mosquitoes. However, European
strains did cause major epidemics in Russia and Poland in the 1920s,
with high death rates as far north as Archangel, Russia. In Holland,
mosquitoes sheltering in the warmth of stables and human habitations
continued to feed and transmit malaria throughout the winter and during
colder summers. Thus, the dormancy and delayed onset of P. vivax
were probably not essential for transmission, and it is conceivable that
P. falciparum was endemic in England during the Little Ice Age.
Alternatively, a more virulent strain of vivax may have been
present. The question may never be resolved because the strains involved
are now extinct.
The Cure
The strongest evidence that ague was indeed malaria is the identity
of its cure. Until the mid-20th century, the only effective remedy for
malaria—at least in western medicine—was an extract of cinchona
powder, obtained from the bark of several tree species native to the
Andes, in South America. The principal active ingredient in this bark is
quinine, a drug that has probably benefited more people than any other
in the combat of infectious disease. Quinine is still used today, but
few people know that its effective use for malaria therapy was first
developed in tests with ague patients living in the salt marshes of
Essex, less than 50 km from the center of London. What is more, the
field experimentation that led to this English remedy was conducted
during the coldest years of the Little Ice Age.
The first prescription of cinchona powder in England is attributed to
Robert Brady in 1660. Thomas Sydenham advocated its use in his Methodus
curandi febres in 1666. By that time, the "Jesuit's Powder"
was already widely known in Europe, but in Protestant England many
orthodox physicians were prejudiced against its use—partly because its
export from Peru and Bolivia was in the hands of Catholics and its use
had not been mentioned in the classical medical texts of Galen and
partly because a reliable prescription had not been developed. In the
end, popularization of the drug came in a highly unorthodox manner: a
relatively untutored man, Robert Talbor, abandoned his apprenticeship to
an apothecary to develop a safe dosage and an effective treatment
regimen: "I planted myself in Essex near the sea side, in a place
where agues are the epidemical diseases, where you will find but few
persons but either are, or have been afflicted with a tedious quartan."
After several years of study and testing, he developed what we would now
call a patent medicine, a secret formulation that was essentially an
infusion of cinchona powder in white wine.
In 1672, Talbor popularized his remedy by publishing Pyretologia: a
Rational Account of the Cause and Cures of Agues. The success of his
treatments became widely known and brought him rapid fame and fortune.
Charles II appointed him Physician Royal in 1672. He was knighted in
1678, after he cured the King of an ague (20). Sir
Robert Talbor then traveled to France, where he cured the son of Louis
XIV. With the additional title of Chevalier Talbot, he became famous
throughout Europe, curing Louis XIV, Louisa Maria, Queen of Spain, and
hundreds of other royal and aristocratic persons (Figure
5).
Talbor's rise to fame has been told in many publications (21,22),
yet none of these mention that the entire story took place in a period
when temperatures were probably colder than in any other period in the
past 10,000 years. Records for central England from 1670 to 1700 suggest
that snow lay on the ground for an average of 20 to 30 days (in some
years more than 100 days) as opposed to 2 to 10 days in the present
century. In the winter 1683-84, the ground was frozen to more than 1 m.
Belts of sea ice 5 km wide were present along the coast in the English
Channel and are believed to have been 30 to 40 km wide off the coast of
the Netherlands. The average summer growing season was approximately 5
weeks shorter than in the 20th century, and in some years the difference
may have been more than 2 months (1). Nevertheless, the
fact that the European aristocracy was eager to pour money and honors on
an untutored commoner suggests that malaria continued to be a serious
problem.
Defoe and Beyond
Robinson Crusoe was shocked to discover that a footprint in the sand
was not his own: "so that I shook with cold, like one in an
ague." His creator, Daniel Defoe (1660-1731), had traveled
extensively in southern England in 1685 to 1690. In A Tour through the
Whole Island of Great Britain (23), he described the
depredations of ague in the Dengie marshes of Essex (Figure
6), an area 70 km east of London that remained notorious for the
disease until the end of the 18th century (15):
". . . our London men of pleasure . . . go from London on purpose
for the pleasure of shooting [the abundant wildfowl] but those gentlemen
who . . . go so far for it, often return with an Essex ague on their
backs, which they find a heavier load than the fowls they have
shot."
These sportsmen may have been wealthy enough to buy Talbor's remedy,
but ordinary people suffered greatly in these unhealthy marshes. They
risked living there because, as the land was excellent for rearing
high-quality sheep and cattle, landlords were willing to pay high wages:
" . . . for it being a place where every body cannot live, those
that venture it, will have encouragement, and indeed it is but
reasonable they should."
However, as in the malarious tropics today, the nonimmune were in
great danger. Thus Defoe observed: "a strange decay of the [female]
sex here . . . it was very frequent to meet with men that had had from
five to six, to fourteen or fifteen wives . . . the reason . . . was
this; that they [the men] being bred in the marshes themselves, and
seasoned to the place, did pretty well with it; but that they always
went into the hilly country . . . for a wife: that when they took the
young lasses out of the wholesome and fresh air, they were healthy,
fresh and clear, and well; but when they came out of their native aire
into the marshes . . . they presently changed their complexion, got an
ague or two, and seldom held it above half a year, or a year at most;
and then . . . [the men] would go to the uplands again, and fetch
another; so that marrying of wives was reckoned a kind of good farm to
them."
During the 18th century, temperatures began to return to pre-mid-16th
century levels. However, the wide variability of the Little Ice Age
continued for at least 150 years. In the 1770s, much as today, alarm
that the climate was becoming increasingly erratic prompted new emphasis
on the recording of weather variables.
There are numerous accounts of malaria in all the northern European
countries in the 18th and early 19th centuries. The wealth of records in
this period confirms that the disease was common at many coastal sites
in England and in some parts of Scotland, with occasional transmission
as far north as Inverness (57°20'). The northern limit (24)
was roughly along the 15°C July isotherm—not the 15°C winter
isotherm, as stated by some authors (8,10).
Thus, there was endemic transmission in southern Sweden and Finland,
with occasional devastating epidemics that extended to the northern end
of the Gulf of Bothnia, close to the Arctic Circle (25,26).
In North America transmission occurred throughout most of the United
States (27) and in some parts of Canada (28).
In 1827, John Macculloch (29) wrote "We may
take the average of life among ourselves, in round numbers, at fifty
with sufficient safety for this purpose. In Holland it is twenty-five;
the half of human life is cut off at one blow, and the executioner is
malaria, for there is no other cause for the superior mortality of that
country."
The same was probably true of the coastal marshes of England,
although the incidence appears to have decreased over the century. After
the 1880s, transmission dropped precipitously, and the disease became
relatively rare, except during a short period following World War I.
This decline cannot be attributed to climate change, for it occurred
during a warming phase, when temperatures were already much higher than
in the Little Ice Age. Factors contributing to the decline include the
following: Mosquito habitat was eliminated by improved drainage and
extensive land reclamation. Root crops, such as turnips and
mangel-wurzels, were introduced as winter fodder, enabling farmers to
maintain much greater numbers of animals throughout the year; this
diverted An. atroparvus, a zoophilic species, from feeding on
humans. Rural populations declined as manual labor was replaced by
machinery, further reducing the availability of humans as hosts for both
mosquitoes and parasites. New building materials and improved methods of
construction made houses more mosquito-proof, especially in winter, thus
reducing the risk for contact with mosquitoes. Greater access to medical
care and a rapid drop in the cost of quinine reduced the survival rate
of the malaria parasite in its human host.
A similar decline occurred in the more prosperous countries of
Europe—Norway, Sweden, Denmark, Germany, Holland, Belgium, France, and
northern Italy. However, malaria maintained a much firmer grip on
Eastern Europe—Finland, Poland and Russia, and the countries bordering
the Black Sea and the eastern Mediterranean. It was not until the advent
of DDT, after World War II, that a concerted attempt could be made to
eradicate the disease from the entire continent (30).
At the same time, the Communicable Disease Center (forerunner of the
Centers for Disease Control and Prevention) was set up in Atlanta to
eliminate malaria from the United States, where it was still endemic in
36 states (27), including Washington, Oregon, Idaho,
Montana, North Dakota, Minnesota, Wisconsin, Iowa, Illinois, Michigan,
Indiana, Ohio, New York, Pennsylvania, and New Jersey.
Conclusion
In 1975, the World Health Organization declared that Europe was free
of malaria. The last indigenous case in England had been in the 1950s
and in Bruegel's Holland in 1961. Results were equally spectacular on
other continents. By 1977, 83% of the world's population was living in
regions from which malaria had been eradicated or control activities
were in progress. The only areas in which the control effort was limited
were those of high endemic stability, particularly sub-Saharan Africa.
However, this momentum could not be sustained, and the goal of
worldwide eradication was abandoned. Today, the disease is again common
in many parts of Central America, the northern half of South America,
much of tropical and subtropical Asia, some Mediterranean countries and
many of the republics once part of the Union of Soviet Socialist
Republics. This rapid recrudescence has been attributed to population
increase, forest clearance, irrigation and other agricultural
activities, ecologic change, movement of people, urbanization,
deterioration of public health services, resistance to insecticides and
antimalarial drugs, deterioration of vector control operations, and
disruptions from war, civil strife, and natural disasters. Claims that
malaria resurgence is due to climate change ignore these realities and
disregard history. For example, the many statements that recent climate
change has caused malaria to ascend to new altitudes (10,31,32)
are contradicted by records of its distribution in 1880 to 1945 (33-35).
With the return of malaria, there has been an exponential rise in
international travel. Tens of thousands of cases are imported into
Europe and North America each year. As was anticipated 30 years ago (36),
a few of these cases give rise to autochthonous transmission by
indigenous mosquitoes.
In much of Western Europe, the likelihood that malaria will become
reestablished is probably small (37). However, in
countries (e.g., in the Balkans) where malaria control has ceased but
the probability of transmission remains high, reintroduction could
threaten the public health. Indigenous transmission associated with
imported cases has recently been reported in Kazakhstan, Kyrgyzstan,
Turkmenistan, Uzbekistan, Bulgaria, the Republic of Moldova, Romania,
Italy, and Corsica, and the malaria-free status of Europe may be in
jeopardy (38). Public concern should focus on ways to
deal with the realities of malaria transmission, rather than on the
weather.
|
|
![]()